
Note: Over a long period of time, the structure of atoms had been developed. Models of the Atom Dalton’s model (1803) Thomson’s plum-pudding model (1897) Rutherford’s model (1909) Bohr’s model (1913) Charge-cloud model (present) Chapter 11 11.1 Rutherford’s Atom: To describe Rutherford’s model of the atom. RUTHERFord's MoDEL 61 2.5.2 The Difficulties. Thus, the solar system model of the atom was not accepted. solar system is the force of gravity while that acting in an atom is the Coulomb force. The much lighter electrons, he assumed, lay well outside the. Thats equivalent in scale to a marble in the middle of a football stadium. This will result in the loss of energy, which causes the accelerated charge to fall inside the nucleus. Instead, in 1911, Rutherford cooked up a new model of the atom in which all of the positive charge is crammed inside a tiny, massive nucleus about ten thousand times smaller than the atom as a whole (Fig 3). An accelerated charge which is an electron, will then emit energy as electromagnetic radiation. Problem 1: In Rutherford’s nuclear model of the atom, the nucleus is analogous to the sun which is about which electrons move around the orbit (radius 10-10 m) like the earth orbits around the sun. Even if they travel at the same velocity in modulus, the trajectory of that velocity varies with time, resulting in centripetal acceleration. (1MeV is 1. Iron-56 (5625Fe) has a binding energy per nucleon of 8.79 MeV. 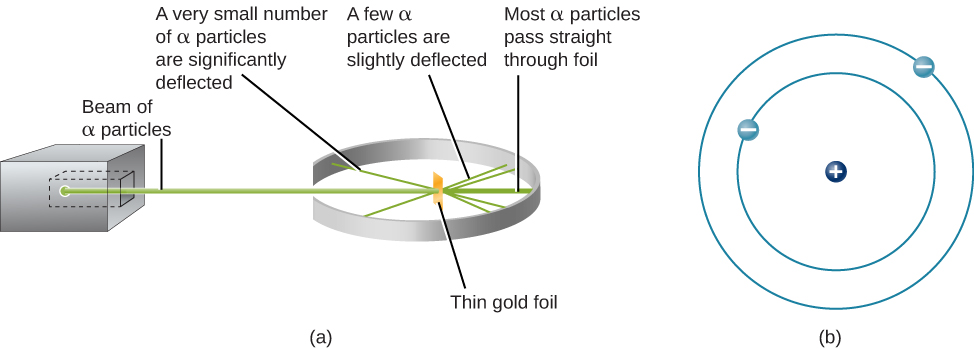
The concern about this model is that electrons are charged particles that have centripetal acceleration while travelling in a circle. How is the degradation of orbiting planets in our solar system like that of the Rutherford atomic model Explain. The “solar system” model represents an atom as a massive positive body which is the nucleus (like the sun in the solar system) with negative entities which are the electrons (like the planets in the solar system) orbiting around it. The majority of an atom is made up of free space. The protons and neutrons are crammed into the nucleus of the atom, while the electrons, which are much smaller, orbit around the outside. Protons, electrons, and neutrons are the elements that make up an atom. Hint:Atoms are the fundamental building blocks of all matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
